Research Highlights
OPRM1/MRGPRX1 heterodimers drive opioid-induced itch through a peripheral mechanism
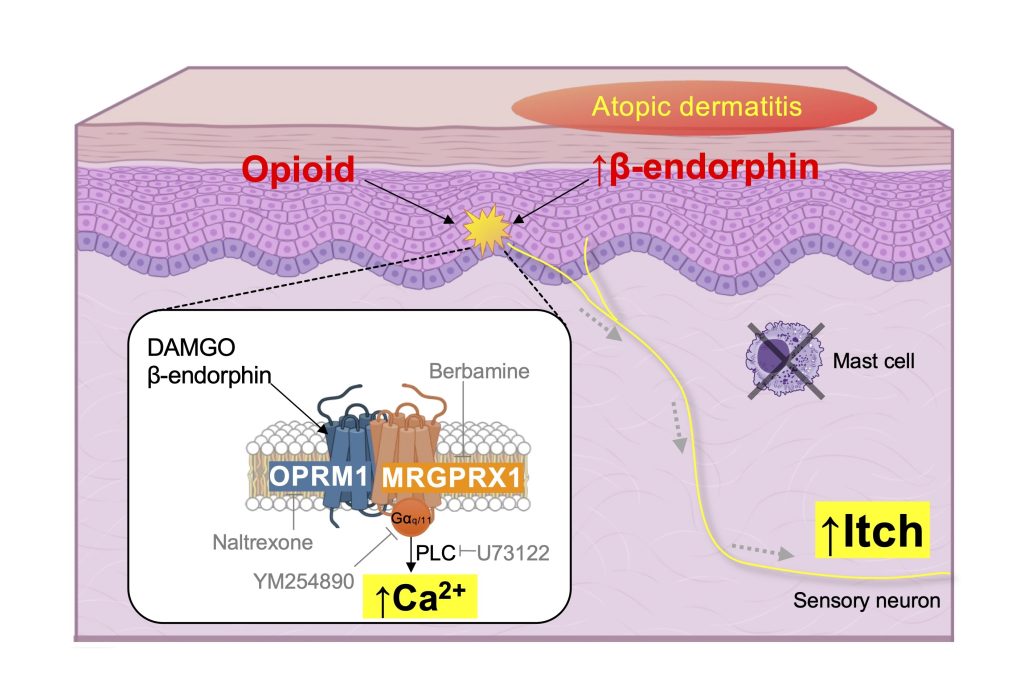
Opioid-induced itch is a common and distressing side effect of opioid analgesics, yet its underlying mechanisms remain poorly understood. While central µ-opioid receptor (OPRM1) signaling has been implicated, emerging evidence suggests that peripheral mechanisms also contribute, although their specific roles have not been clearly defined. We investigated the interaction between OPRM1 and the itch-specific receptor MRGPRX1 in sensory neurons using bimolecular fluorescence complementation (BiFC), calcium and cAMP imaging, siRNA knockdown, and pharmacological inhibition assays. Behavioral assays in mice were conducted to assess scratching responses. We also employed immunohistochemistry, RT-qPCR, and ELISA to evaluate gene and protein expression levels in dorsal root ganglia (DRG) and skin tissues, including a mouse model of atopic dermatitis (AD). OPRM1 formed heterodimers with MRGPRX1 in HEK293T cells and sensory neurons, triggering a signaling switch from Gαi/o-mediated cAMP inhibition to Gαq/11-driven calcium mobilization upon activation with DAMGO or endogenous opioids. This heterodimerization elicited robust intracellular calcium responses and scratching behavior in mice, which were attenuated by OPRM1 or MRGPRX1 antagonists. In the AD mouse model, increased OPRM1 expression and β-endorphin levels were observed in DRG neurons, correlating with heightened scratching and calcium responses. In contrast, although the δ-opioid receptor (OPRD1) associated with MRGPRX2, it did not trigger mast cell degranulation, suggesting a limited contribution to peripheral itch signaling. Our findings identify a novel peripheral mechanism of opioid-induced itch mediated by OPRM1/MRGPRX1 heterodimers in sensory neurons. This receptor complex promotes calcium signaling and itch behavior, distinct from central or mast cell-dependent pathways. Targeting this heterodimer may offer new therapeutic strategies to alleviate opioid-induced itch without impairing analgesia.
Sanjel B, Rawal D, Kim MR, Lee WJ, Jeong KW, Shim WS.
J Biomed Sci. 2026 Mar 28;33(1):35.
https://doi.org/10.1186/s12929-026-01238-x
Azelaic acid potentiates TRPV3 activity as a mechanism for skin irritation
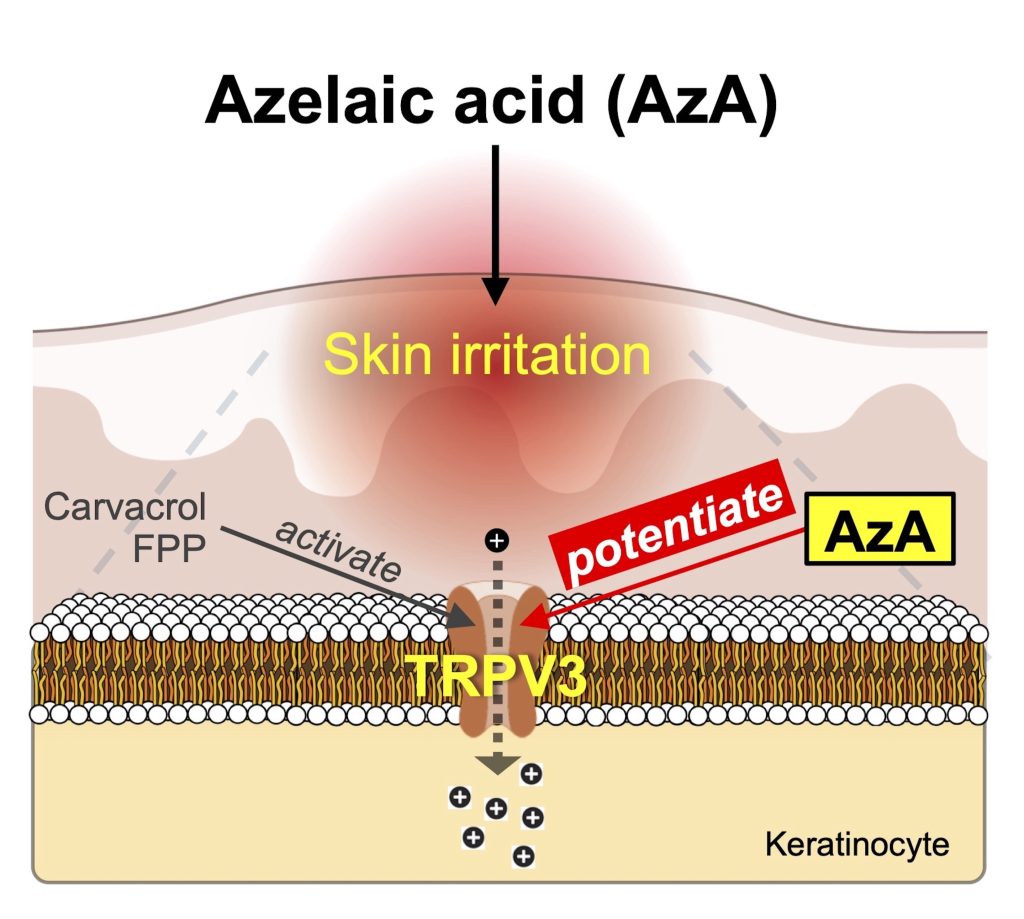
Azelaic acid (AzA), a naturally occurring dicarboxylic acid used for treating acne and rosacea, is generally well tolerated but often causes skin irritation. However, the underlying molecular mechanisms remain unclear. Here, we investigated whether AzA modulates sensory ion channels or receptors to induce these effects. Computational prediction using ReverseDock and DrugBAN identified the thermosensitive TRPV3 channel, highly expressed in keratinocytes, as a candidate AzA-interacting protein. Calcium imaging in HEK293T and HaCaT cells showed that AzA does not directly activate TRPV3 but markedly potentiates its responses to agonists such as carvacrol and endogenous farnesyl pyrophosphate (FPP). Docking and mutagenesis studies pinpointed Tyr565 as critical for this effect. In vivo, AzA enhanced TRPV3 dependent scratching behavior and calcium responses in mouse keratinocytes. Repeated exposure led to skin lesions, upregulation of Trpv3 and farnesyl diphosphate synthase (Fdps), suggesting elevated endogenous FPP synthesis. TRPV3 antagonists significantly reduced these effects, confirming specificity. Similar potentiation was observed in human keratinocytes. Collectively, our findings demonstrate that AzA functions as a modulator of TRPV3, enhancing sensitivity to endogenous and exogenous agonists. This mechanism provides a molecular explanation for AzA-induced irritation and suggests strategies to minimize side effects while retaining therapeutic benefits.
Rawal D, Lee WJ, Shim WS. Azelaic acid potentiates TRPV3 activity as a mechanism for skin irritation.
J Invest Dermatol. 2026 Feb 3:S0022-202X(26)00071-0.
https://doi.org/10.1016/j.jid.2026.01.022
Vitexin alleviates atopic dermatitis-associated itch via TRPV4 inhibition in sensory neurons and MRGPRX2/MrgprB2 blockade in mast cells
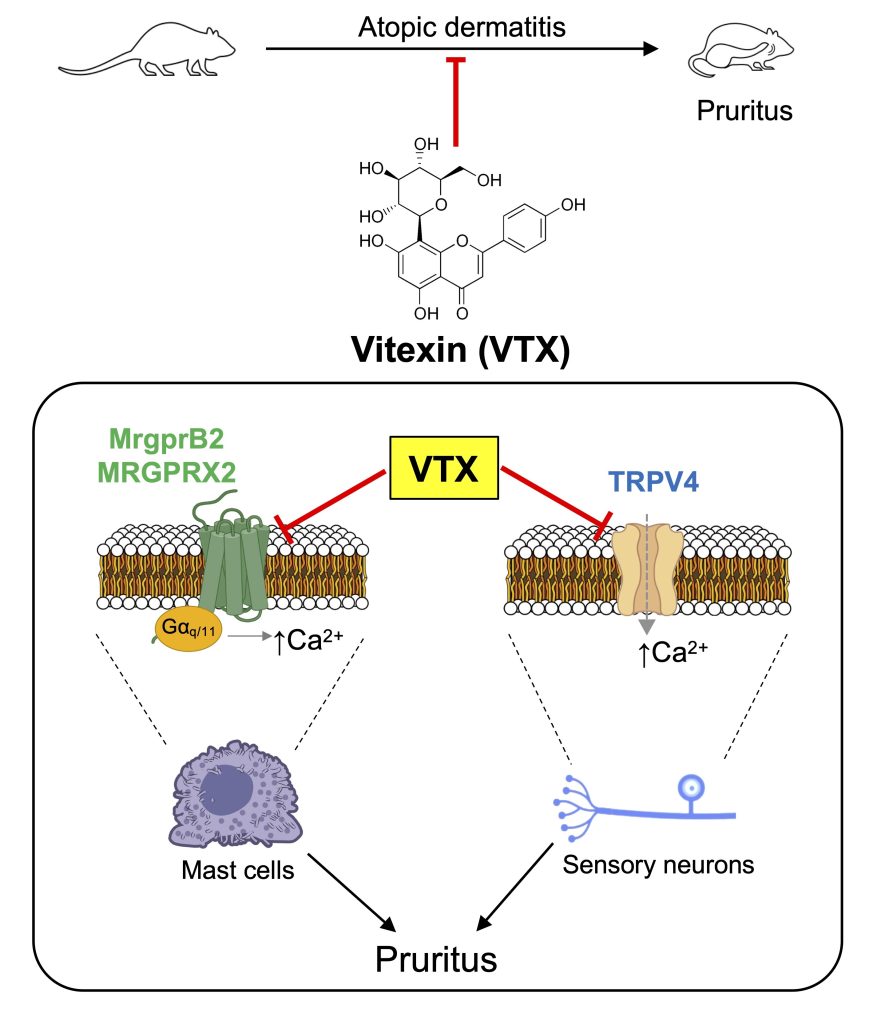
Chronic itch is a debilitating symptom in atopic dermatitis (AD), often resistant to current antipruritic therapies. Here, we demonstrate that vitexin (VTX) exerts antipruritic effects through modulation of neuronal and mast cell pathways. Calcium imaging in TRPV4-transfected HEK293T cells and primary mouse dorsal root ganglion neurons demonstrated that VTX substantially inhibited TRPV4-dependent calcium influx, reducing both peak responses and the proportion of agonist-responsive neurons. Parallel assays in HEK293T cells expressing human MRGPRX2 and murine MrgprB2 revealed dose-dependent suppression of compound 48/80-induced calcium signaling. Mast-cell degranulation assays indicated that VTX markedly decreased β-hexosaminidase release from primary mouse peritoneal mast cells and HMC1.2 cells upon compound 48/80 stimulation. In silico molecular docking analysis showed that VTX occupied overlapping binding regions with known agonists in TRPV4 and MRGPRX2/MrgprB2, suggesting direct receptor interactions. In an MC903-induced murine model of AD, VTX reduced scratching behavior, mast cell activation, and transcriptional upregulation of Trpv4 and MrgprB2. Notably, antipruritic effects were enhanced in Trpv4-deficient mice, indicating the contribution of MRGPRX2/MrgprB2 suppression. These findings identify VTX as a promising antipruritic candidate for AD-associated pruritus, and mechanistic insights provide a rationale for further exploration of VTX in advanced AD models and eventual translation to clinical therapies.
Kim Y, Choi J, Kim M, Rawal D, Lee WJ, Shim WS. Vitexin alleviates atopic dermatitis-associated itch via TRPV4 inhibition in sensory neurons and MRGPRX2/MrgprB2 blockade in mast cells.
Int Immunopharmacol. 2026;168(Pt 1):115757.
doi:10.1016/j.intimp.2025.115757
Sphingosylphosphorylcholine induces itch via activation of TRPM3 and TRPA1 in mice
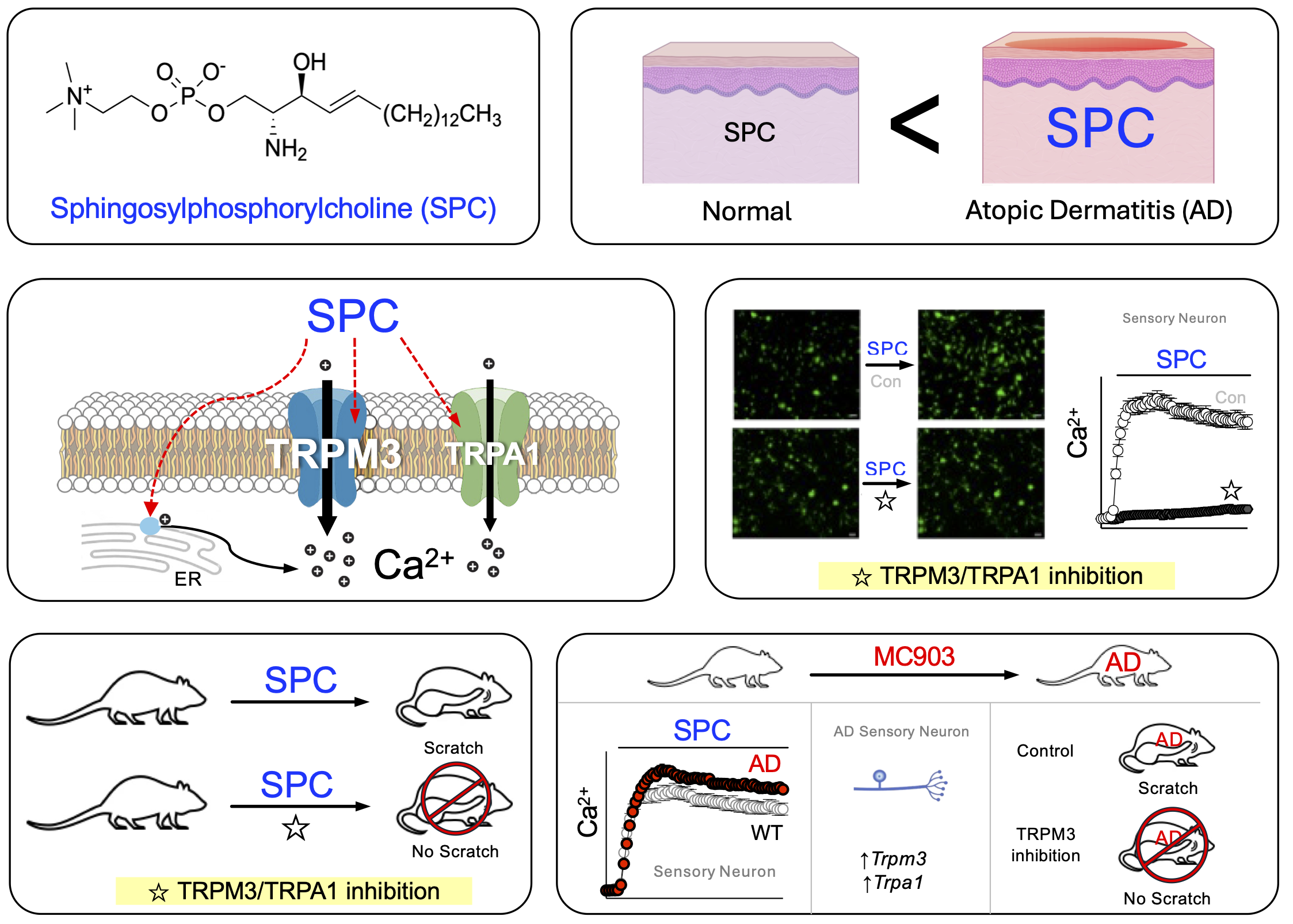
Itch is a prevalent symptom in atopic dermatitis (AD), often leading to a strong urge to scratch. Elevated levels of sphingosylphosphorylcholine (SPC) are found in the stratum corneum of AD patients, and while SPC is known to induce itch, its molecular targets are not well understood. This study aims to identify the signaling pathway of SPC-induced itch under AD conditions. We demonstrate that SPC specifically activates the Transient Receptor Potential Melastatin 3 (TRPM3) channel in sensory neurons. In HEK293T cells expressing TRPM3, SPC treatment caused a significant increase in intracellular calcium, which was inhibited by TRPM3 antagonists. Among various TRP channels tested, TRPM3 exhibited the highest reactivity to SPC, followed by TRPA1. Molecular docking analysis also supported interactions between SPC and both TRPM3 and TRPA1. In an AD mouse model, SPC-induced responses were dependent on TRPM3 and TRPA1, and the expression of these channels increased in dorsal root ganglion neurons. SPC-induced scratching behaviors were significantly reduced by TRPM3 and TRPA1 antagonists, with TRPM3 playing a critical role in spontaneous scratching. This study identifies TRPM3 and TRPA1 as key mediators of SPC-induced itch, providing potential therapeutic targets for treating itch in AD patients.
Song DE, Rawal D, Lee WJ, Shim WS. Sphingosylphosphorylcholine induces itch via activation of TRPM3 and TRPA1 in mice.
Biochem Pharmacol. 2025 Jul;237:116952.
https://doi.org/10.1016/j.bcp.2025.116952
Glucosylsphingosine evokes pruritus via activation of 5-HT2A receptor and TRPV4 in sensory neurons.
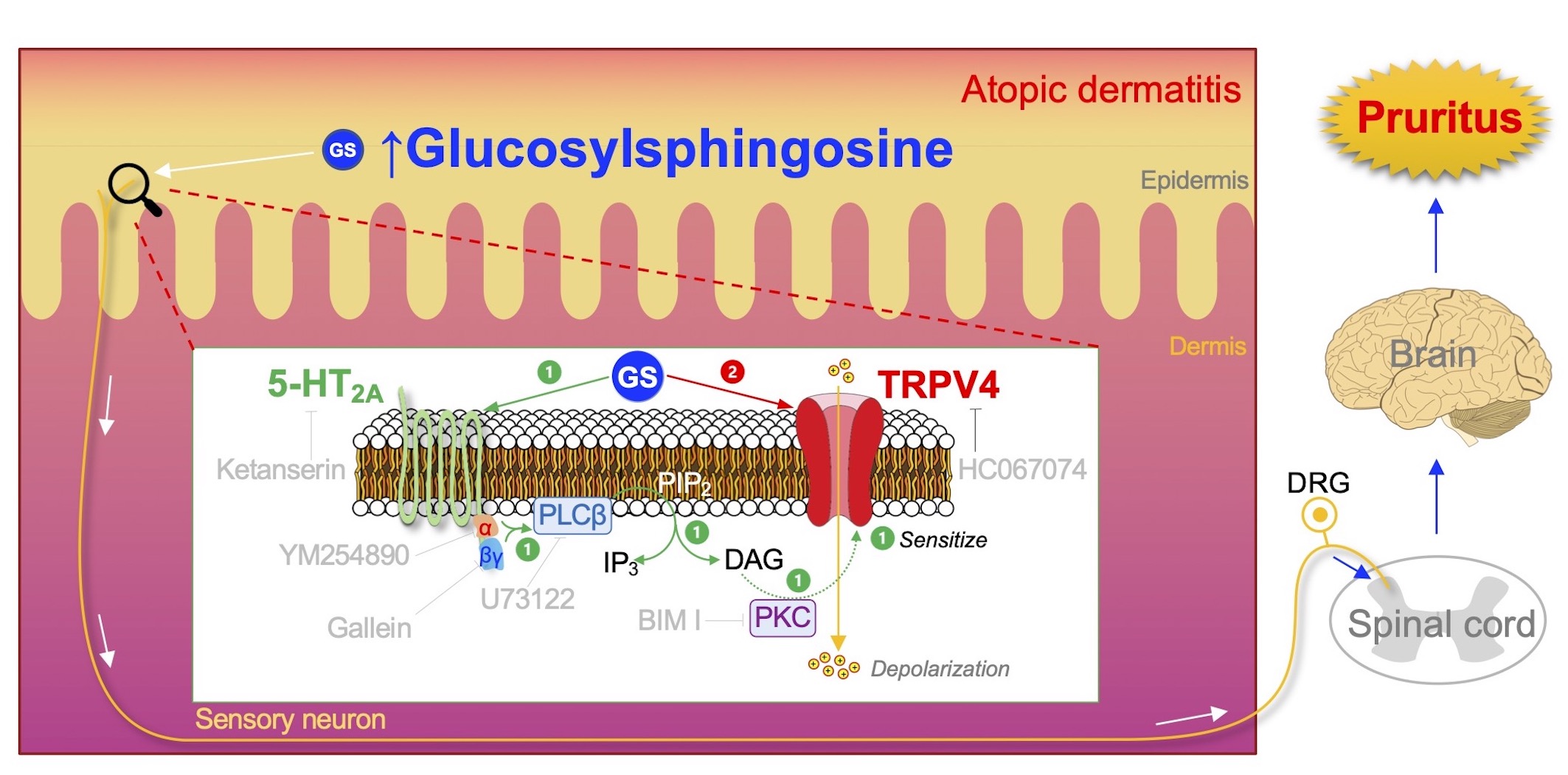
The highly accumulated GS in the epidermis of patients with AD will activate the peripheral sensory neuron to induce pruritus in the following way: (1) GS activates 5-HT2A receptor (5-HT2A) with subsequent stimulation of PLC via Gαq/11 and Gβγ complex, which in turn breaks down phosphatidylinositol 4,5-bisphosphate (PIP2) into inositol triphosphate (IP3) and diacylglycerol (DAG). DAG will then activate phosphokinase C (PKC), which leads to sensitization of TRPV4. (2) Activation of the sensitized TRPV4 can be directly facilitated by GS, which will allow a massive influx of cations from the extracellular regions, increasing a chance to induce depolarization and possibly an action potential. This GS-induced electrical signal produced in the peripheral sensory neuron will be further transmitted through the spinal cord and to the brain, where the signal can finally be perceived as pruritus.
Sanjel B, Kim BH, Song MH, Carstens E, Shim WS. Glucosylsphingosine evokes pruritus via activation of 5-HT2A receptor and TRPV4 in sensory neurons.
Br J Pharmacol. 2022 May;179(10):2193-2207.
https://doi.org/10.1111/bph.15733
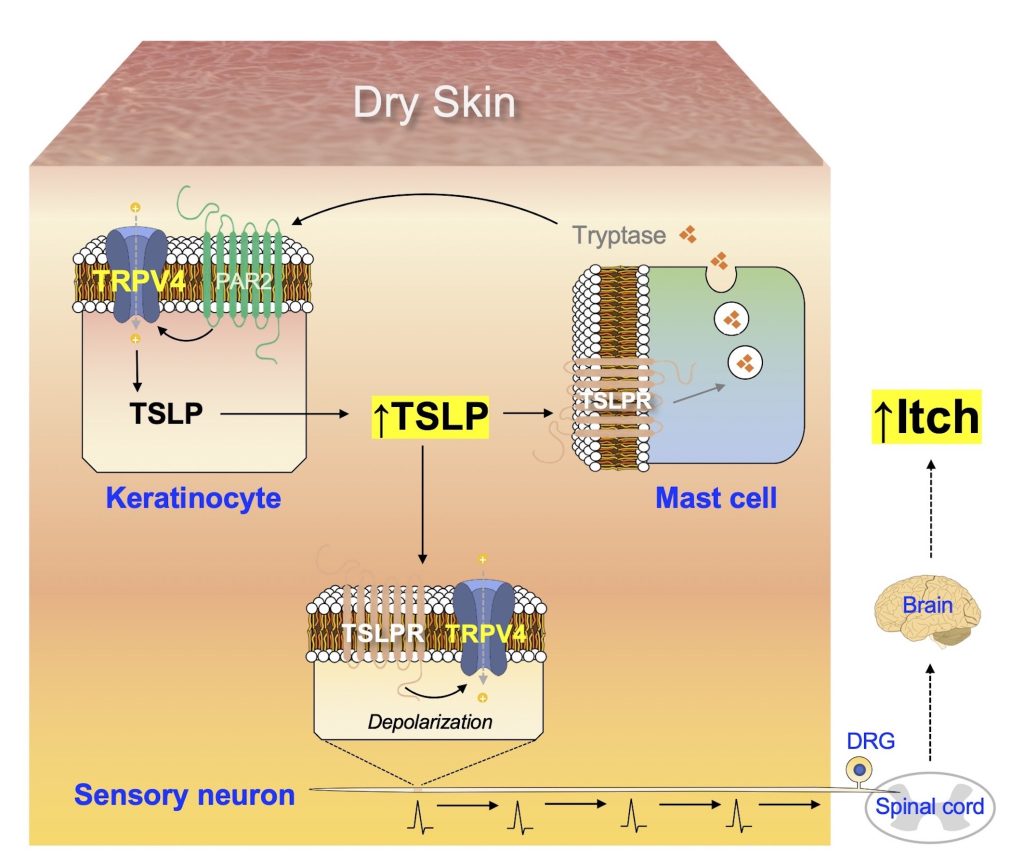
Cutaneous Neuroimmune Interactions of TSLP and TRPV4 Play Pivotal Roles in Dry Skin-Induced Pruritus
The production of TSLP in keratinocytes increases under dry skin conditions, in a TRPV4-dependent manner. Additionally, TSLP acts on TSLPR in mast cells to promote the release of tryptase, which in turn stimulates the keratinocytes via PAR2 and TRPV4 to produce more TSLP, thus generating a positive feedback loop of increased TSLP production. TSLP can also act on TSLPR in sensory neurons to transmit electrical signals towards the spinal cord, which are conveyed to the brain and finally perceived as itch.
Lee WJ, Shim WS. Cutaneous Neuroimmune Interactions of TSLP and TRPV4 Play Pivotal Roles in Dry Skin-Induced Pruritus.
Front Immunol. 2021 Dec 2;12:772941.
Caffeic acid phenethyl ester inhibits pseudo‑allergic reactions via inhibition of MRGPRX2/MrgprB2‑dependent mast cell degranulation
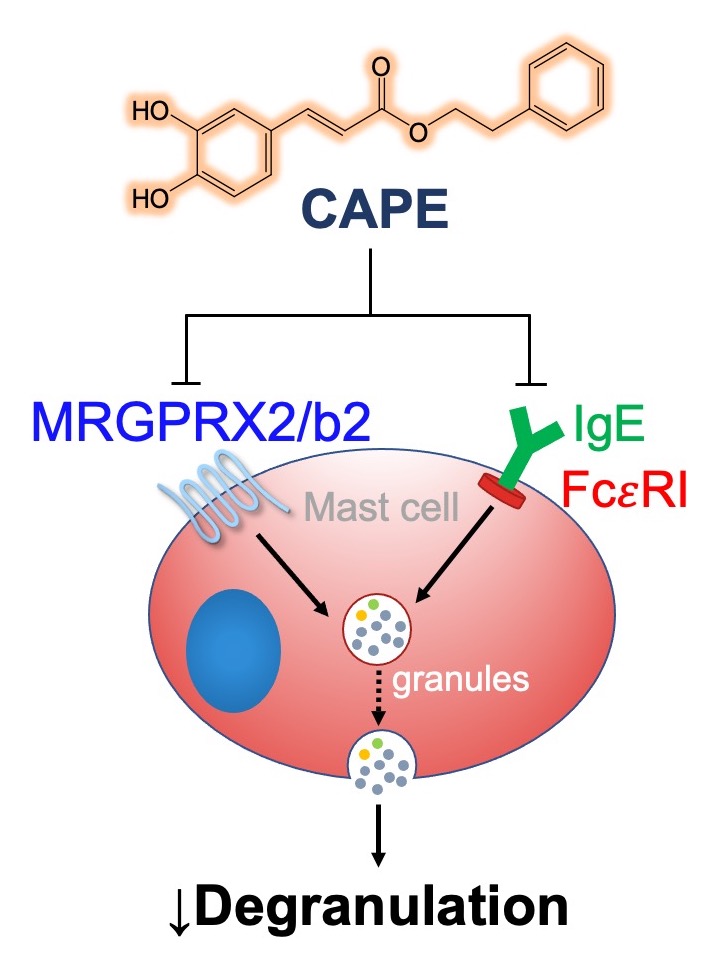
Mast cells play essential role in allergic reactions through the process called mast cell degranulation. Recent studies have found that a basic secretagogue compound 48/80 (C48/80) induces non-IgE-mediated mast cell degranulation via activation of human Mas-related G protein-coupled receptor X2 (MRGPRX2) and mouse MrgprB2. Although previous studies have revealed that caffeic acid (CA) and its derivatives possess anti-allergic effects via IgE-dependent manner, it is largely elusive whether these compounds have impact on MRGPRX2/MrgprB2 to exert inhibitory effects. Therefore, the present study investigated whether CA as well as its derivatives – rosmarinic acid (RA) and caffeic acid phenethyl ester (CAPE) – has the ability to inhibit the activity of MRGPRX2/MrgprB2 to evoke pseudo-allergic effects. As a result, it was found that CAPE inhibits C48/80-induced activation of MRGPRX2/MrgprB2, but neither CA nor RA showed discernible inhibition. Furthermore, the β-hexosaminidase release assay showed that CAPE inhibits mouse peritoneal mast cell degranulation in both IgE-dependent and MrgprB2-dependent manners. Additionally, mouse paw edema induced by C48/80 was dramatically suppressed by co-treatment of CAPE, suggesting that CAPE possesses a protective effect on C48/80-evoked pseudo-allergic reactions. The pretreatment of CAPE also significantly decreased scratching bouts of mice evoked by C48/80, demonstrating that CAPE also has an anti-pruritic effect. Therefore, these data implicate that CAPE can suppress pseudo-allergic reactions evoked by C48/80 via MrgprB2-dependent manner. Finally, molecular docking analysis showed that CAPE is predicted to bind to human MRGPRX2 in the region where C48/80 also binds, implying that CAPE can be a competitive inhibitor of MRGPRX2. In conclusion, it is found that CAPE has the ability to inhibit MRGPRX2/MrgprB2, leading to the prevention of mast cell degranulation and further to the alleviation of mast cell reactions. These results indicate that CAPE as a CA derivative could be developed as a new protective agent that exerts dual inhibition of mast cell degranulation mediated by IgE and MRGPRX2/MrgprB2.
Adhikari N, Shim WS. Caffeic acid phenethyl ester inhibits pseudo-allergic reactions via inhibition of MRGPRX2/MrgprB2-dependent mast cell degranulation.
Arch Pharm Res. 2022 Sep;45(9):644-657
https://doi.org/10.1007/s12272-022-01405-2
